Summary.
The RAAS is a critical hormonal cascade that helps maintain arterial blood pressure, extracellular fluid volume, and electrolyte balance. It co-ordinates renal, vascular, and adrenal functions and interfaces with the sympathetic nervous system, vasopressin release, natriuretic peptides and local tissue systems (notably in the heart, kidney, and brain). It is a finely tuned, multi-organ system that maintains cardiovascular and fluid homeostasis.
Dysregulation contributes to hypertension, heart failure, kidney disease, and inflammatory pathologies. The RAAS is a critical hormonal cascade that helps maintain arterial blood pressure, intravascular volume, and electrolyte balance. It co-ordinates renal, vascular, and adrenal functions and interfaces with the sympathetic nervous system, vasopressin release, and local tissue systems (notably in the heart, kidney, and brain).
Modern therapeutics target this system at multiple points — renin, ACE, AT₁ receptors, and aldosterone — to modulate its activity and prevent end-organ damage.
Pathway.
Renin is synthesised and stored in juxtaglomerular (JG) cells of the kidney afferent arteriole, and is released into the blood in response to reduced renal perfusion pressure (detected by juxtaglomerular baroreceptors) and to reduced sodium chloride delivery to the macula densa.
It is also released in response to sympathetic activation via β₁-adrenoceptors at the juxtaglomerular cells.
Renin, an aspartyl protease, cleaves angiotensinogen in plasma to the decapeptide angiotensin I (Ang I).
Angiotensinogen, itself, is a glycoprotein produced mainly by the liver. Its production is increased by oestrogen, corticosteroids, thyroid hormone, and inflammation (IL-6–mediated).
Circulating angiotensin I is inactive but is converted to the active angiotensin II by Angiotensin-Converting Enzyme (ACE) on the vascular endothelial cells - particularly in the pulmonary and renal circulations. Angiotensin-Converting Enzyme removes two amino acids from angiotensin I to form angiotensin II (Ang II). It also degrades bradykinin, a vasodilator peptide, adding to the vasoconstrictive effect .
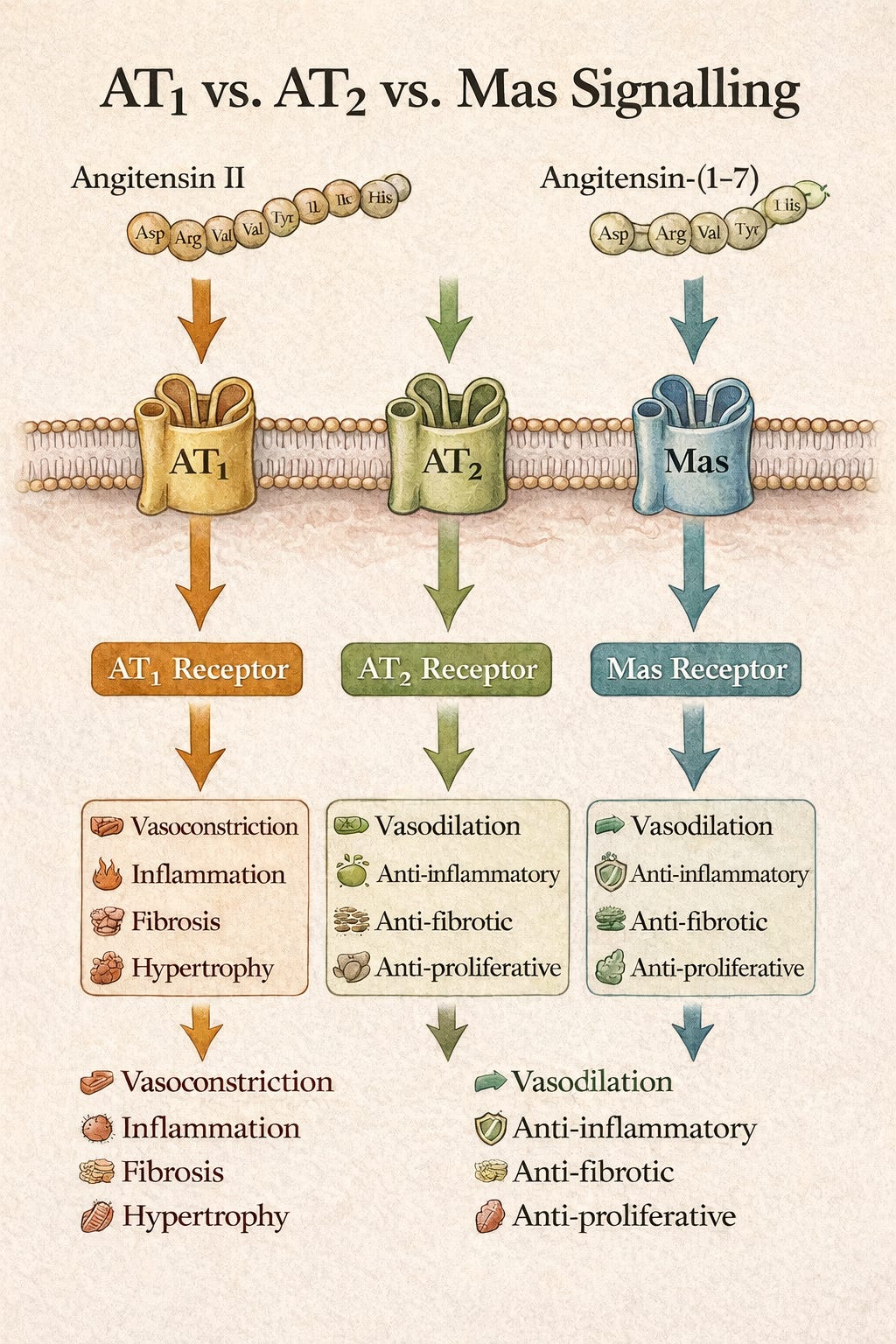
Angiotensin II acts mainly on the angiotensin II receptors AT₁ and AT₂ (both G protein-coupled receptors).
AT₁R: Mediates vasoconstriction, aldosterone release, sympathetic activation, sodium retention, and cellular growth.
AT₂R: Counter-regulatory — promotes vasodilation, anti-proliferation, apoptosis, and NO release. Although angiotensin II binds both AT₁ and AT₂ receptors, many of the vasodilatory and anti-fibrotic effects classically attributed to AT₂ activation are mediated in parallel by the angiotensin-(1–7)/Mas receptor axis.
Aldosterone is synthesised in the zona glomerulosa of the adrenal cortex. It is released following Ang II stimulation, directly in hyperkalaemia and, to a lesser extent, in response to ACTH.
It binds mineralocorticoid receptors in the distal nephron (principal cells) to:
↑ Na⁺ reabsorption via the epithelial sodium channels (ENaC) channels.
↑ K⁺ secretion via the renal outer medullary potassium (ROMK) channels.
↑ water reabsorption (secondary to sodium retention).
Aldosterone activity results in expanded extracellular volume and elevated blood pressure.
The effects of RAAS activity are counterbalanced by ACE2 which converts Ang II to Ang (1–7). This acts via Mas receptors producing vasodilation, anti-fibrosis, and anti-inflammatory effects.
Natriuretic peptides (ANP, BNP) also oppose RAAS activity by promoting loss of sodium in the urine, vasodilation, and suppression of renin/aldosterone.
Physiological Effects.
Cardiovascular.
Angiotensin II acts directly upon AT₁ receptors in vascular smooth muscle producing vasospasm, so increasing systemic vascular resistance (SVR). If the cardiac output remains constant, this action will increase the blood pressure.
Direct cardiac effects include positive inotropy and myocardial hypertrophy. Chronic stimulation can lead to myocardial fibrosis.
Renal.
RAAS activity promotes efferent arteriolar constriction which maintains glomerular filtration pressure during hypotension.
At the renal tubules, aldosterone increases Na⁺ reabsorption and K⁺ secretion in the principal cells of the distal tubule and collecting duct via its effect upon the epithelial sodium channels (ENaC) and the renal outer medullary potassium channels (ROMK channels).
Neurohormonal.
The RAAS can stimulate thirst through its effects upon the hypothalamus, and increase ADH (vasopressin) release which leads to water retention.
There is also sympathetic activation via brainstem nuclei, with consequent adrenergic effects upon the heart and circulation.
Cellular Mechanisms.
AT₁ Receptor: A G-protein–coupled receptor (GPCR) activating phospholipase C leads to IP₃ + DAG, ↑ intracellular Ca²⁺ and, consequently, vasoconstriction, aldosterone synthesis, and cellular growth.
Aldosterone mechanism: Nuclear receptor binding leads to increased transcription of ENaC and Na⁺/K⁺-ATPase subunits so facilitating increased Na⁺ re-uptake.
Clinical Relevance.
In heart failure, chronic RAAS activation leads to vasoconstriction, sodium retention and cardiac remodelling. ACE inhibitors, ARBs, MRAs (spironolactone) can be beneficial.
Hypertension is associated with over-activity of the angiotensin II/aldosterone axis. Appropriate therapeutic options include ACE inhibitors, angiotensin receptor blockers and renin inhibitors.
Glomerular hypertension and fibrosis from persistent from persistent angiotensin II stimulus is a feature of chronic kidney disease (CKD). RAAS blockade can slow CKD progression.
In COVID-19, use of ACE2 receptor by the coronavirus disrupts balance between angiotensin II and angiotensin (1-7) and AT₁R and MasR signalling - a possible link to endothelial injury and cytokine storm; also to potential therapeutic options.
Suggested Reading.
Classical RAAS physiology
Hall JE, Guyton AC
Textbook of Medical Physiology
Elsevier (latest ed.)
Gold-standard integrative physiology reference
Chapters on renal sodium handling, RAAS, pressure–natriuresis
Especially strong on system-level control of BP
https://www.elsevier.com/books/guyton-and-hall-textbook-of-medical-physiology/hall/978-0-323-59712-8
Atlas SA
The renin–angiotensin aldosterone system: pathophysiological role and pharmacologic inhibition
Journal of Managed Care Pharmacy (2007)
Clear overview of classical RAAS
Good bridge between physiology and therapeutics
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8673245/
DOI: https://doi.org/10.18553/jmcp.2007.13.s8-b.9
Peach MJ
Renin–angiotensin system: biochemistry and mechanisms of action
Physiological Reviews (1977)
Older but still authoritative
Excellent mechanistic detail on Ang II signalling
https://journals.physiology.org/doi/10.1152/physrev.1977.57.2.313
ACE2, Ang-(1–7), Mas receptor (counter-regulatory RAAS)
Donoghue M et al.
A Novel Angiotensin-Converting Enzyme–Related Carboxypeptidase (ACE2) Converts Angiotensin I to Angiotensin 1-9
Circulation Research Volume 87, Issue 5, 1 September 2000; Pages e1-e9
Landmark paper identifying ACE2 as a functional RAAS enzyme
https://doi.org/10.1161/01.RES.87.5.e1
Santos RAS, et al.
The ACE2/Angiotensin-(1–7)/Mas Axis of the Renin–Angiotensin System: focus on angiotensin-(1–7)
Physiological Reviews, 2018
This review highlights roles of Ang-(1–7) via the Mas receptor and describes how this axis counterbalances the “classical” RAAS (Ang II/AT$_1$) in the heart, vessels, and kidneys.
https://pubmed.ncbi.nlm.nih.gov/29351514/
DOI: https://doi.org/10.1152/physrev.00023.2016
Patel VB, Zhong J-C, Grant MB, Oudit GY
Role of the ACE2/Ang-(1–7) Axis of the Renin-Angiotensin System in Heart Failure
Circulation Research (2016)
Clinical relevance of ACE2
Heart failure, vascular disease, remodelling
https://www.ahajournals.org/doi/10.1161/CIRCRESAHA.116.307708
DOI: https://doi.org/10.1161/CIRCRESAHA.116.307708
RAAS, ACE2, and modern disease (HFpEF, lung, endothelium)
Imai Y, Kuba K, Rao S et al.
Angiotensin-converting enzyme 2 protects from severe acute lung failure
Nature (2005)
Key demonstration of ACE2 as a protective enzyme
Foundational for later COVID-19 work
https://www.nature.com/articles/nature03712
DOI: https://doi.org/10.1038/nature03712
Vaduganathan M et al.
Renin–angiotensin–aldosterone system inhibitors in patients with COVID-19
NEJM (2020)
Clinical clarification of ACEi/ARB effects on ACE2
Important for modern interpretation